Metropolitan AntiViral Drug Accelerator
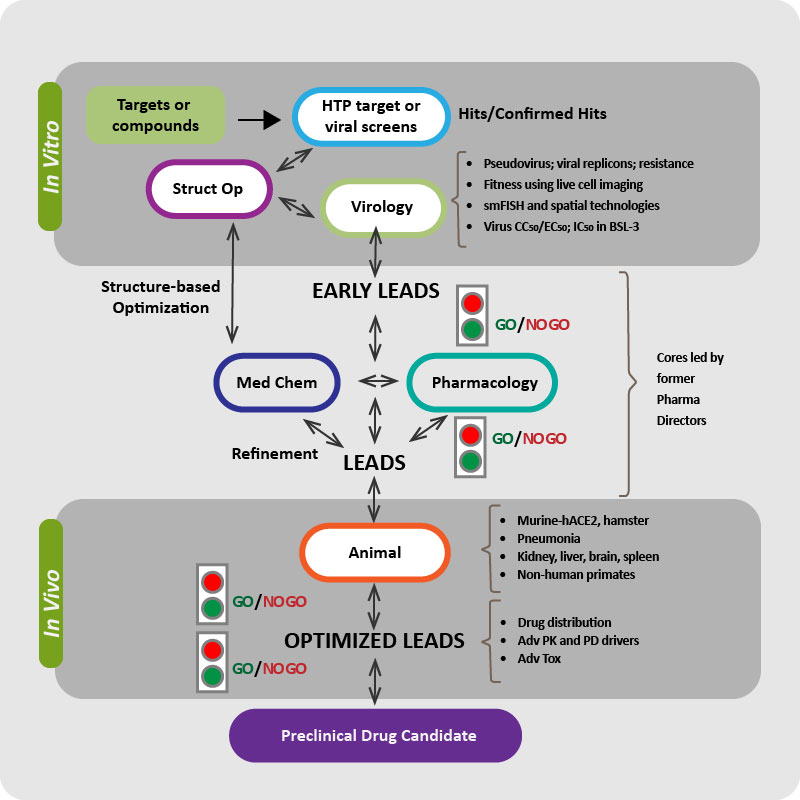
The mission of the Metropolitan AntiViral Drug Accelerator (MAVDA) is to conduct innovative, multidisciplinary research to develop, optimize and test innovative small molecule antiviral drugs that target coronaviruses (such as SARS-CoV-2), and other viral families with high potential to cause a pandemic in the future. Our goal is to rapidly develop antiviral drugs that can be given orally in an outpatient setting.
MAVDA is funded by a three-year, $65,141,731 million grant from the National Institutes of Health (NIH) and the National Institute of Allergy and Infectious Disease (NIAID). MAVDA is one among the nine Antiviral Drug Discovery (AViDD) Centers for Pathogens of Pandemic Concern program funded by the NIAID, and involves world-class institutions in the New Jersey-New York metropolitan area, and beyond.
For more information on AViDD and the program, visit the program’s website. MAVDA is principally located in New York City and New Jersey. It consists of academic and 3 commercial partners.
Developmental Research Projects (DRPs) are intended to expand the scope and range of current research and investigators involved in antiviral discovery, allowing for testing of novel ideas (with limited preliminary data) and development of new technologies.
Mentored Research Projects (MRPs) are intended to increase the availability of qualified researchers and other personnel for antiviral discovery research by providing opportunities to further their professional advancement. MRPs must relate to the Center objectives and may be used to support post-doctoral fellows, early career investigators, or senior investigators new to the field of antiviral discovery and development.